Understanding Clinical Trials
For Patients
Clinical trials are scientific studies designed to test new and improved ways to treat and manage cancer. In order to understand how clinical trials may benefit you, it is important to appreciate how they work.
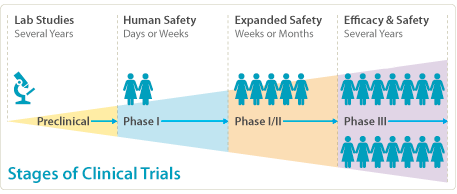
Cancer clinical trials are conducted in three different phases
Each phase occurs one after another, if the one before is proven to be successful. Different questions about the new treatment are answered with each phase.
Phase I
- The first study that involves people
- Looks at how a new treatment should be given (e.g. by mouth, IV drip, or injection?), how often (e.g. how many times a day?) and, if it involves medication, what dose safe is safe
- Only involves a small number of people
Phase II
- Continues to test the safety of the treatment
- Assesses how well the new treatment seems to work (e.g. Does the tumour shrink? Does it improve blood test results?)
- As in Phase 1 only involves a small number of people
Phase III
- Compares a new treatment to the best standard treatment, to work out which is best (e.g. which group has better survival rates? Fewer side effects?)
- Assigns people at random to receive the new treatment or the best standard treatment
- May involve hundreds or thousands of people from around the state, country or world
Randomised Controlled Trials
A controlled trial compares a new treatment with the current best standard treatment (the control). In a randomised controlled trial, some people receive the new treatment while others receive the standard treatment.
Treatment will be chosen by a computer which randomly assigns people to each group. This is done so each group has a similar mix of people- to ensure the people getting each treatment can be compared without bias. Neither you nor your doctor can choose which treatment you will receive.
Benefits of clinical trials
Clinical trials contribute to knowledge and progress towards treating cancer. For many patients, clinical trials may be a potential option to access experimental and cutting edge therapy. If a new treatment proves effective in a study, it may become a new standard treatment that can help future patients.
Because of progress made through clinical trials, many people treated for cancer are now living longer, and experience less side effects from their treatment.


